Stopping aHUS treatment when no longer needed is a big decision for patients. Global Action put the patients research agenda question to our expert.
Can a complement inhibitor be stopped safely when not needed by some aHUS patients and what makes them different?
This was the response
“The decision to stop or adjust complement inhibition in atypical hemolytic uremic syndrome (aHUS) patients should be made in consultation with healthcare professionals and is based on the individual’s specific medical circumstances. While some patients may be able to safely discontinue complement inhibitors when they are no longer needed, it’s important to consider several factors that can make individuals with aHUS different:
- Genetic Mutations: The underlying genetic mutations associated with aHUS can vary from person to person. Some individuals may have genetic mutations that lead to ongoing complement system dysregulation, making continued complement inhibition necessary.
- Triggers: aHUS episodes can be triggered by various factors, including infections, medications, pregnancy, or other stressors. Patients who experience aHUS due to such triggers may be able to discontinue complement inhibitors once the trigger is removed or resolved.
- Response to Treatment: The effectiveness of complement inhibition can differ among aHUS patients. Some individuals may respond well to treatment and experience disease remission, while others may require ongoing therapy for disease management.
- Regular Monitoring: Ongoing monitoring of aHUS patients is essential to assess their response to treatment and the stability of their condition. Healthcare professionals can make treatment adjustments based on individual patient needs.
- Pediatric Patients: Treatment considerations and the potential for discontinuation may differ for pediatric aHUS patients. Their care should be tailored to their specific circumstances.
The decision to discontinue complement inhibitors should be made with caution and under the guidance of healthcare professionals who have experience in managing aHUS. Regular follow-up appointments and ongoing monitoring are crucial to evaluate the need for continued treatment and to ensure patient safety.”
Global Action supports safe stopping of treatment when the clinical evidence shows it is not needed. The stopping process should have safety built into it to avoid any patient harm, including an assured and rapid return to treatment pathway if needed.
Several major international stopping treatment studies, including STOPECU , CURIHUS , the Milan Clinic have found that stopping treatment can be done. The studies have led to greater understanding of which patient might’ go into a lasting remission and which might relapse too quickly . In 2024 the SETS study will report on not only its stopping treatment outcomes but also the safety of the way it was done.
So the answer to stopping “YES”, and “ Yes , some understanding” of the patient differences is emerging. Of greater importance is the safety of the way it is done and that is yet to be fully established and practiced, so watch this space!
Article No. 625
Some of the other stopping treatment articles on Global Action’s website.
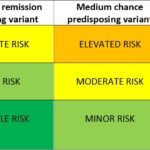
aHUS – Stopping eculizumab treatment safely
For aHUS patients stopping eculizumab (or even Ravulizumab) treatment is an important but emotive issue. Having had the treatment when seriously ill and then recovered, no one wants to go…
Withdrawal evidence patients need to know
Article No. 400 5 December 2020 Whilst on the topic of safe withdrawal from eculizumab treatment ( see previous articles 398 and 399), further evidence was published just a month…
CONTINUE READINGWithdrawal evidence patients need to know
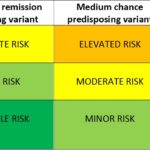
Eculizumab withdrawal risk profiling
Article No. 398 4 December 2020 In its previous article “Dip into ASH 2020” Global Action highlighted research results relating to “aHUS” and “TMA” which are to be presented at…
CONTINUE READINGEculizumab withdrawal risk profiling
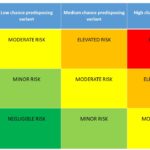
RISK OF WITHDRAWAL OF ECULIZUMAB
In the current aHUS related trials list reported on recently, there were two which were looking at the safe withdrawal of eculizumab. In addition to these two trials, it is…

