Sometimes there are fascinating articles related to atypical HUS which people might miss, and it’s then that it is particularly helpful to have the aHUS global action team direct our spotlight on aHUS research. Such is the case during this first half of 2021, when research into COVID-19 infection includes insights into inflammation, complement activation, and thrombotic microangiopathy (damage from tiny clots in small blood vessels throughout the body). This new information does more than lead to a better understanding of COVID-19 issues and care, since expanded knowledge about the complement system (part of the body’s immune response) touches upon several key issues related to aHUS.
People who are genetically predisposed to aHUS may experience their first episode of disease early or quite late in life, with potential multi-organ damage from symptoms that can vary greatly in severity and duration. There is still much unknown about how certain circumstances regarding genetics and environmental factors may combine to trigger atypical HUS disease activity, such as: infections (bacterial/viral), pregnancy, certain drugs, organ transplantation, cancer, and inflammation or chronic/autoimmune disease impact. As research progresses, perhaps we’ll also gain more understanding of ‘idiopathic’ cases – people diagnosed with atypical HUS who do not appear to have any underlying cause or genetic factors.
Research articles about atypical HUS are listed within our website on topic-specific lists within our Info Centre, where you can see a list for “New Research” which are then also listed by category. In addition updating our scrolling lists regarding research into aHUS treatment and so on, we’ve added an entirely new list “Discontinuing Treatment / Relapse” to reflect information now available on studies into stopping treatment, optimal treatment duration, and risk of aHUS relapse with treatment withdrawal.
We’d like to highlight here a few recent additions to the research and publications page, a resource created and maintained by the aHUS Alliance Global Action team. Click here to visit this virtual ‘library’ specific to Atypical HUS Research & Publications
Garred et al. Therapeutic Targeting of the Complement System: From Rare Diseases to Pandemics
In atypical HUS damage often occurs to the kidneys, and sometimes other major organs, when part of the body’s immune system (complement) becomes over-activated. This article contains sections on ‘Complement Attack in Tissue Damage’ and the ‘Role of an Uncontrolled Overactivated System’. As such, broad interest in complement may lead to expanded drug discovery.
Why do those interested in aHUS keep mentioning complement, to include drugs that are classified as ‘complement inhibitors’? The first line of this publication’s abstract provides this brief history: “The complement system was discovered at the end of the 19th century as a heat-labile plasma component that “complemented” the antibodies in killing microbes, hence the name “complement.”
Merinero et al. Functional characterization of 105 Factor H variants associated with atypical HUS: lessons for variant classification
While still new (29 June 2021) with only its abstract publicly available, this publication hits several points of high interest to aHUS patients and those who treat them. The aHUS Global Patients’ Agenda stated research questions that patients and their families want answered, and interestingly those aspects below were also central questions within Merinero team’s article as well. Paraphrased from the aHUS Global Patients’ Agenda, you’ll find such questions listed as agenda items 1.1, 1.2, and 3.1:
What are the predisposing genetic & triggering factors for, and how variable are the risks among individuals?
On issues of genetic testing for aHUS patients and their family members, what are the implications and what can be done with that knowledge?
Is it more clinically effective, as well as cost effective, to undertake genetic testing to help guide & manage aHUS patient’s treatment?
Given the genetic component involved with atypical HUS, people are understandably concerned with passing along a genetic predisposition for aHUS to their children. Pregnancy can trigger aHUS activity, during gestation but also peri-partum (time immediately surrounding the birth) and well into post-partum (after delivery). While these below are listed as ‘New Research’ on our aHUS Research & Publications page, many more articles are listed within topic-specific categories to include: Pregnancy, Genetics, and Complement.
Ardissino et al. Risk of Atypical HUS Among Family Members of Patients Carrying Complement Regulatory Gene Abnormality
Its title nicely portrays the focus of this publication, and with a conclusion touches upon genetic testing, pregnancy, and risk.
“In conclusion, the risk of developing the disease in any given relative of a patient carrying complement regulatory gene mutations responsible for aHUS in a family member can be estimated to be 20%, thus lower than the reported 50%. This information, which is missing in the current literature, can be very important for patient’s relatives who understandably wonder whether they or their children may also develop this severe life-threatening disease. Although the penetrance is not as high as previously reported, in our opinion the severity of the condition may justify the screening of relatives for the specific mutation responsible for the disease in their family. The test being relatively inexpensive, we tend to screen subjects based on their willingness to be aware of their specific risk (particularly in case of mutations involving C3 or CFH genes in the index case, multiple cases in the same family, young brothers and siblings of the index case, and no clear trigger in the index case). The awareness of the risk can be very important in specific settings (peripartum or in case of severe triggering diseases or major surgeries) where the timing of treatment may be crucial for a better outcome or even for surviving this life-threatening disease.”
While a 2020 release, the publication from Timmermans et al titled The natural course of pregnancies in women with primary atypical haemolytic uraemic syndrome and asymptomatic relatives also points to similar interests in family genetics for aHUS patients of child-bearing age. This study analyzed “maternal and fetal outcomes of 39 pregnancies from 17 women with primary aHUS and two asymptomatic relatives.” and noted diagnostic difficulties which exist regarding preeclampsia and HELLP syndrome. Pregnancy-related TMA can cause a variety of complications for women with atypical HUS, as noted in the publication by Gunay et al in March, with the self-explanatory title Pregnancy-related atypical hemolytic uremic syndrome with renal, cardiac and obstetric complications and a satisfactory recovery: a case report
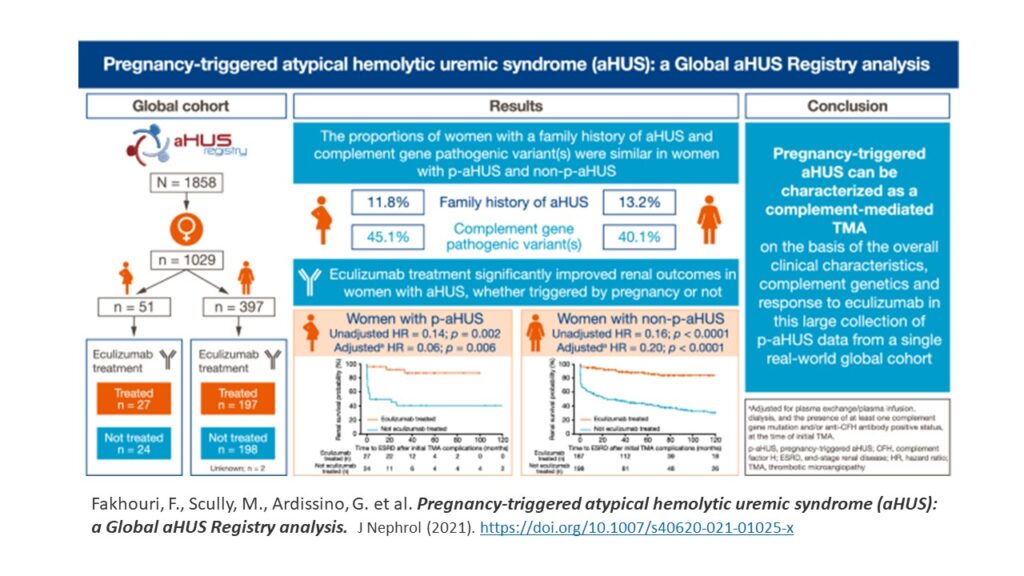
Fakhouri et al. Pregnancy-triggered atypical hemolytic uremic syndrome (aHUS): a Global aHUS Registry analysis
With data from a large and international group of women with pregnancy-associated atypical HUS (p-aHUS), this publication from the Global aHUS Registry concluded:
“Findings from this large collection of data from a single, real-world, global cohort confirm that pregnancy-triggered aHUS is comparable to aHUS with no identified trigger. Our results indicate that pregnancy-triggered aHUS is not only a disease of the postpartum period, and that presumed pregnancy-associated TMAs may well be aHUS. The similarities in demographics, clinical characteristics, complement genetics, disease progression, and response to eculizumab between women in both groups confirm that pregnancy-triggered aHUS can be considered a complement-mediated TMA.”
Here’s an infographic from that study
The much-anticipated group of research into how long to treat, and assessing risk for relapse, has been published during the past several months. It’s clear from titles, such as Brodsky’s Eculizumab and aHUS: to Stop or Not that there is no clear answer on optimal duration of treatment. (Note: Brodsky’s article regards the Fakhouri et al research noted below). At present there is not a clear set of guidelines and standardized methods to monitor aHUS patients who withdraw from treatment. While Brambilla et al (2020) noted the role of urine dip strips for home monitoring to help identify early stages of aHUS relapse, there seems some differences between eculizumab and ravulizumab when measuring effectiveness of complement inhibition as determined by free C5 and CH50 complement assays (Cataland et al, 2019). Of more recent note regarding aHUS research on treatment withdrawal and risk factors:
Chaturvedi et al. Outcomes of a clinician-directed protocol for discontinuation of complement inhibition therapy in atypical hemolytic uremic syndrome
No longer considered a life-long drug, eculizumab withdrawal issues are varied.
“In summary, we show that for most aHUS patients, discontinuation of C5 inhibitor (eculizumab) therapy is safe and acceptable without loss in renal function when done under close monitoring to allow early detection and treatment of relapses. Multicenter, prospective studies are needed to establish the renal and vascular safety of limited duration eculizumab therapy for aHUS, which has the potential to reduce the financial burden, the inconvenience of frequent infusions, and the small but real risk of meningococcal infection associated with indefinite therapy with terminal complement inhibitors.”
This study mentioned risk of relapse and noted as an key point at the opening of the article, “CFH and MCP variants may be associated with higher risk of relapse, which needs to be evaluated in larger, multicenter studies.”
While the pandemic slowed in-person meetings and delayed the start of some clinical trials, we expect to see a robust fall expansion of studies related to aHUS. We admire and applaud the commitment of clinicians and research teams who have carried on their work despite the restrictions imposed by COVID-19.
ISN has continued its excellent and robust agenda of online nephrology webinars for “MedEd” and new information related to aHUS topics will be presented at upcoming medical society meetings such as Kidney Week (ASN, early Nov 2021) and the 63rd ASH meeting in December for the hematology community. There is usually quite a bit of news surrounding research and clinical trial updates at events occuring in November and December, and our aHUS research scrolling lists will be updated accordingly.
Are you staying current with aHUS research & News?
We invite you to subscribe to our free newsletter, The aHUS Global Advocate
For More about our Newsletter & reasons to Stay Connected
Click Here to Sign Up to receive the Newsletter