Answers to the RDD2022 video continue.
Samantha asks will there ever be a cure for aHUS?
Crikey that is a difficult one to answer. The simplest answer is “I don’t know” , But then nothing is truly impossible, is it Samantha?

100 years ago Complement had been discovered. But back then doctors did not know what our disease was? If they did, they would not have known how to treat it. What happens today would have seemed like science fiction to them then.
100 years from this time , aHUS patients, if there are any, will likely be saying it was obvious what could have been done once we developed “X”.
But first what would a cure be? A cure is different from being in remission although an aHUS patient would feel no different. In remission a patient would face the prospect that there could be another episode at some time, or maybe not, for the rest of their lives. There is no back of the mind certainty. With a cure the prospect of relapse disappears altogether.
For a cure to happen, whatever it is each individual aHUS patient has about them which can make their own Complement damage the linings of their own blood vessels, and that cannot stop it from doing so, would have to be fixed. Then it would be cured.
Whether it is something in how Complement components are made by the body, or other parts of the body are made which can also temporarily hamper good Complement from being controlled. There would have to be multiple cures.
For most part Complement is good to have but it can go bad.
But if some components do not work properly then Complement goes haywire and does the same to the body’s own cells. Destroying them, which then leads to the body sending in its clotting system to repair it. But it does not stop the Complement destruction and blood vessels fill up with clots until red blood cannot get through without bursting. And aHUS begins when those cells are in the kidney and it becomes impaired and fails.
It is not the case of having a method of correcting one component of Complement nor just one of the different parts that make up that component. There are many and as mentioned earlier, it is also defects in other parts of the body that have to be dealt with too in some people.
The defects are mutations, or significant variants in the DNA coding that each individual acquires at conception. If the DNA sequence is right then no problem, but if there is a change it could make a difference to how that person’s Complement works.
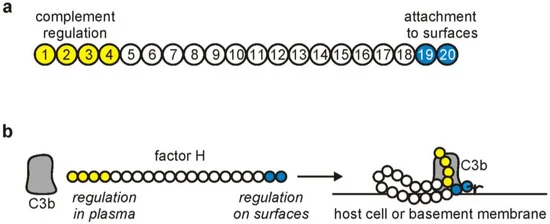
Take this mutation in one part of someone’ Complement, facor H , or CFH , a component which is frequently the cause of aHUS because it cannot do what it is supposed to and without it Complement remains out of control and aHUS results.
c.3643C>G; p. Arg1215Gly
The 20 parts of CFH , or short consensus repeats (SCR), as they are called are illustrated in the diagram below. There are two ends that are important when it comes to aHUS. One end regulates Complement in circulating plasma using parts 1 to 4 , the other end regulates it at blood vessel surfaces , parts 19 and 20. Regulation at blood vessel surface is essential to stop aHUS from happening.
The above variant affects the ability of this person’s CFH SCR 20 to work because the “Arg” , Arginine, part of the DNA has been switched to ”Gly”, Glycine, at conception in one of this persons ancestors and it has now been inherited. To make it work again the “Gly” would have to be genetically modified back to “Arg”. This would have to be done in the liver where CFH is produced.
This variant is just one of hundreds among Complement components that have been found to make someone predisposed to aHUS. Many more are possible as new ones are found each year.

It is not known whether there is any research going on to find a method to change Complement DNA , Israel? If there is no one is saying so. But some such research on Complement genetics is plausible to gain more understanding about it.
Just as Ashley has found has happened for Leukocyte Adhesion Deficiency Type 1 , or LAD 1, a very rare disease like aHUS , which manifests in children. It is an immunodeficiency disorder with an incidence rate of about 1 per million per year. It is caused by mutations in something called ITGB2 which encodes something called CD18. About 90 mutations have been found there. As a result of the mutations patients’ white blood cells are not effective and so LAD 1 patients become more commonly affected by serious infections like peritonitis and pneumonia which are life threatening.

The only cure at present is a haematopoietic stem cell bone marrow transplant. Something used to cure leukaemia, and also PNH (which is treated with eculizumab). Such transplants are quite risky and gruelling procedures.
Gene therapy for LAD 1 involves the cloning of a patients defective stem cell and introducing a healthy copy and then putting it back into the patient’s blood to grow non mutated ITGB2 CD18, so that white blood cells work normally.
And a cure.
More information about the LAD 1 UCLA research can be viewed HERE.
Will this kind of therapy translate to aHUS ? This technology is for blood forming stem cells , most aHUS components are formed in the liver, so something different would be required, Ashley. But maybe one day that something called “X” will be found.
Article No. 494
Complement database leader interview
Professor Stephen Perkins began his research career having studied Chemistry and Molecular Biophysics at Oxford University as well as undertaking Research Fellowships in Switzerland. He moved to University College London…CONTINUE READING

